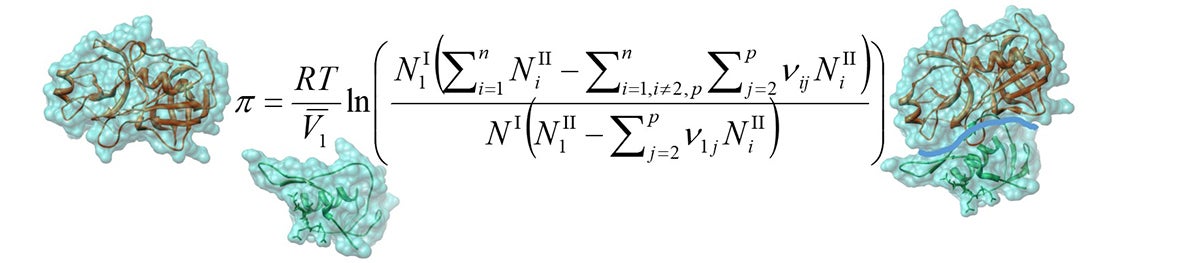Thermodynamics of Crowded Proteins
Overview
We are interested in modeling the behavior of concentrated multicomponent protein (crowded) solutions. Crowded protein solutions are found naturally in cells and have tremendous impact on cellular function. In addition, bioseparations processes often must separate proteinaceous solutions to produce high quality pharmaceuticals.
In general, protein solutions exhibit highly non-ideal thermodynamic behavior. Until now, it was largely accepted that this non-ideality was due to only protein-protein interaction. However, we have shown that, in many cases, hydration and ion binding account for the non-idealities for single, highly concentrated, protein solutions at physiological conditions. In this regard, we have developed a free-solvent based model that applies the related observable parameters to predict crowded protein thermodynamics. Applying the more general form of this model to binary proteins also results in an improved representation of measured osmotic pressure for mixed proteins in solution. Our current research investigates how the osmotic pressure of crowded solutions can provide predictions for protein aggregation and kinetics, which are highly important in our understanding of phenomena that takes place in cellular systems.
Related Publications
Hale CS, McBride DW, Batarseh R, Hughey J, Vang K, Rodgers VGJ (2019) Development and Applications of a Concentrating Membrane Osmometer for Colloid Solutions, Review of Scientific Instruments, 90 (3), 034102.
Hale CS, Ornelas DN, Yang JS, Chang L, Vang K, Batarseh RN, Ozaki N, Rodgers VGJ (2018) Interrogating the Osmotic Pressure of Self-Crowded Bovine Serum Albumin Solutions: Implications of Specific Monovalent Anion Effects Relative to the Hofmeister Series, The Journal of Physical Chemistry B 2018 122 (33), 8037-8046. DOI: 10.1021/acs.jpcb.8b0700.
McBride DW, Rodgers VGJ (2014) A Generalized Free-Solvent Model for the Osmotic Pressure of Multi Component Solutions Containing Protein-Protein Interactions, Mathematical Biosciences, 253:72-87.
McBride DW, Rodgers VGJ (2013) Predicting the Activity Coefficients of Water for Concentrated Globular Proteins Solutions Using Independently Determined Physical Parameters, PLoS ONE, PONE-D-13-35625R1 10.1371/journal.pone.0081933.
McBride D, Rodgers VGJ (2013) Interpretation of Negative Second Virial Coefficients from Non-Attractive Protein Solution Osmotic Pressure Data: An Alternate Perspective, Biophysical Chemistry, Volume 184, 31 December 2013, Pages 79–86, DOI 10.1016/j.bpc.2013.09.005.
McBride D, Rodgers VGJ, (2012) Obtaining Protein Solvent Accessible Surface Area (SASA) Using Osmotic Pressure, AIChE Journal, 58(4):1012-1017, DOI 10.1002/aic.12648.
Yousef MA, Datta R, Rodgers VGJ, (2002) Monolayer Hydration Governs Nonideality in Osmotic Pressure of Protein Solutions”, AIChE Journal, 48(6) 1301-1308.
Yousef MA, Datta R, Rodgers VGJ (2002) Model of Osmotic Pressure for High Concentrated Binary Protein Solutions”, AIChE Journal, 48(4), 913-917.
Yousef MA, Datta R, Rodgers VGJ (2001) Confirmation of Free-Solvent Model Assumptions in Predicting the Osmotic Pressure of Concentrated Globular Proteins”, Journal of Colloid and Interface Science 243 321-325.
Yousef MA, Datta R, Rodgers VGJ (1998) Understanding Non-Idealities of the Osmotic Pressure of Concentrated Bovine Serum Albumin, Journal of Colloid and Interface Science, 207(2), 273-282.
Yousef MA, Datta R, Rodgers VGJ (1998) Free-Solvent Model of Osmotic Pressure Revisited. Application to Concentrated IgG Solution at Physiological Conditions, Journal of Colloid and Interface Science, 197, 108-118.